
Go with confidence
Continuity of care beyond the hospital
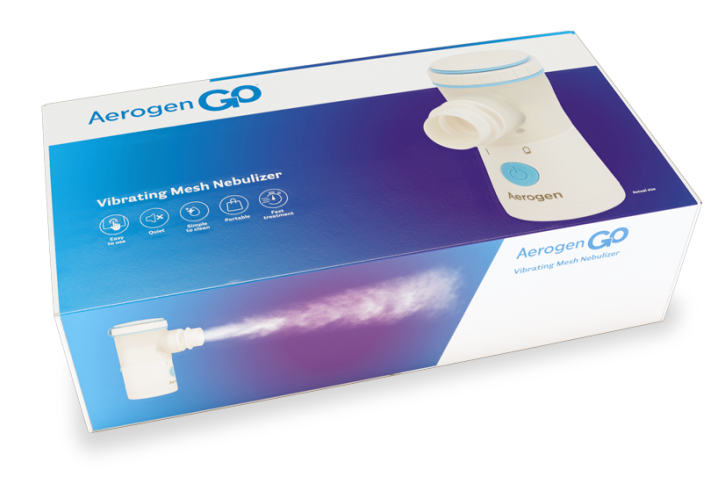
Experience the freedom of the Aerogen Go Vibrating Mesh Nebulizer – a compact, portable*1 vibrating mesh nebuliser for use in hospital, at home, school or while on the go.
Could your respiratory patients benefit from home nebuliser technology?
For patients with respiratory conditions, changes in nebuliser technology from hospital to home can be challenging:
- GOLD guidelines recommend against switching device type in the absence of clinical justification2 to help avoid device confusion.
- Nebulisers that are large, bulky, non-portable or difficult to use can hinder patients’ ability to consistently administer their treatment.3,4
-22e43r.png)
Aerogen Go supports the transition from hospital to home nebuliser therapy
Aerogen Go can be used in adults and children (aged >6 months) to nebulise approved medicines for respiratory treatment.†1
Hospital-quality care in the comfort of a patient’s home.1

Portable, quiet and simple to use,*1 to help support patient adherence to treatment.

Simple and intuitive one-button design.*1
Patient and caregiver surveys have demonstrated satisfaction with nebulised therapy at home and positive patient-reported health outcomes.5,6
Nebulisers may be easier to use than inhalers for some patients, as they require minimal strength, dexterity, and coordination.7-9
(Most patients surveyed had COPD)


Discover seamless care
A single pathway…
A compact and easy-to-use vibrating mesh nebuliser offering continuum of care from hospital to home*1



Aerogen Go can be used by children over the age of 6 months
who require inhalation treatment and can self-ventilate.†1
Aerogen Go comes with the experience and support of Aerogen, the world leader in acute care aerosol drug delivery.‡10
Discover the Aerogen Go® Vibrating Mesh Nebulizer
With the Aerogen Go, patients can confidently manage the treatment of their respiratory symptoms on the go.1
To find out more, and for detailed guidance on the intended use, setup, and cleaning of the Aerogen Go, consult the Instructions for Use.

Enquiries
The Aerogen Go team and our representatives are available to answer your questions and place orders.‡
‡Aerogen Go is currently only available in certain countries.
Manufactured by HCmed Innovations, in compliance with applicable medical device regulations. For further details, please refer to the product Instructions for Use.
GL-3251-2-EN Copyright © 2026 Aerogen Ltd. All rights reserved. AerICU, Aerogen, Aerogen Go, Aerogen Solo, Aerogen Ultra, Go, The WheeZees and Vibronic are trademarks or registered trademarks of Aerogen Ltd or its affiliates.
*Noise level: ≤50 decibels during use. Portability/Size: Dimensions 74 mm x 46 mm x 96 mm; can be battery operated; weight approximately 75 g (without batteries). Ease of use: 2-part design for ease of assembly. Can be used with normal breathing patterns. Battery life: Requires 2 x 1.5V AA alkaline batteries. Batteries may vary among different brands or types of batteries. Keeping spare batteries available is recommended. IP-55 rated: resistant to limited dust ingress and protected against low-pressure water jets from any direction; not designed for full water immersion. Cleaning: 7-step cleaning process after each use and disinfection at the end of each day. Fast treatment delivery: Aerosol output rate: 0.7±0.07 mL/min (2 mL NaF 2.5%).1
†The Aerogen Go® Vibrating Mesh Nebulizer is intended to nebulise approved medication for respiratory treatment. The nebulising system can be used in children over the age of 6 months who require inhalation treatment and can self-ventilate. Children or anyone requiring assistance or supervision must be observed constantly by an adult, caregiver or medical professional when using the device for inhalation therapy. Aerogen Go is designed for single-patient use only.1
‡The world leader in acute care aerosol drug delivery with a global market in 80+ countries.10
COPD, chronic obstructive pulmonary disease; GOLD, Global Initiative for Chronic Obstructive Lung Disease; HF, high-flow; IMV, invasive mechanical ventilation; NaF, sodium fluoride; NIV, non-invasive ventilation; HF, high-flow; SV, self-ventilating.
1. 30-1273 Rev 01 Aerogen Go EU Instructions for Use.
2. GOLD report 2025. Available at: https://goldcopd.org/2025-gold-report/ Accessed: April 2025.
3. Alhaddad B, et al. BMJ Open Respir Res. 2015;2(1):e000076.
4. Sivadasan S, et al. J Pharm Technol. 2021;37(5):254-259.
5. Dumra H, et al. Int J Chron Obstruct Pulmon Dis. 2022;17:2277-2288.
6. Barta SK, et al. Respir Med. 2002;96(6):375-381.
7. Barrons R, et al. Am J Health Syst Pharm 2011;68(13):1221-1232.
8. Talwar D, et al. Indian Chest Society. 2021;38(2):168-173.
9. Geller DE. Respir Care 2005;50(10):1313-1321.
10. Aerogen Data on File.











